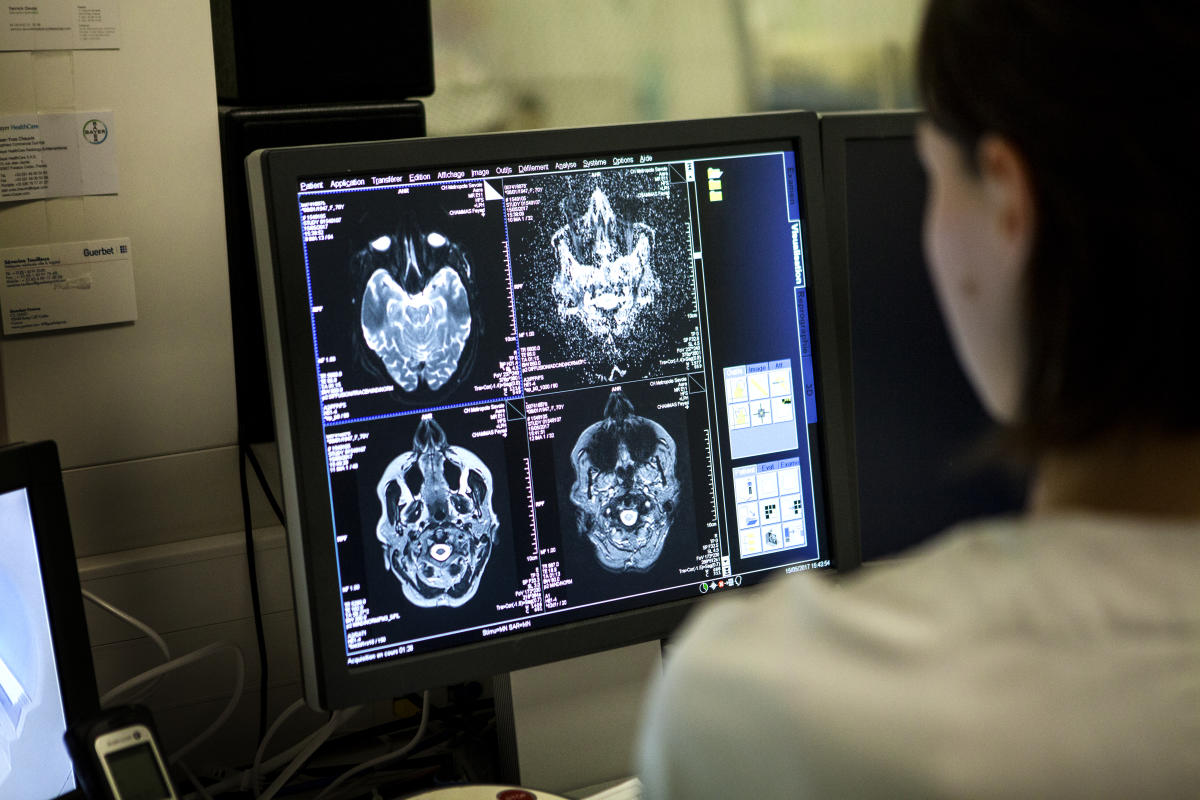
According to reports, the US government has approved software that predicts memory loss based on artificial intelligence for the first time. Darmiyan, a San Francisco-based brain imaging analytics company, he says FDA granted De Novo approval for BrainSee product. According to the medical company, the software platform assigns “an objective score that predicts the likelihood of progression from aMCI to Alzheimer’s dementia within 5 years.” Violent Biotechnology first informed announcement
Darmiyan says BrainSee can predict the progression of memory loss using clinical brain MRI and cognitive tests, which are already standard for patients worried about the first signs of decline. After analyzing imaging and cognitive assessments, the software assigns a predictive score that indicates the patient’s likelihood of memory deterioration over the next five years. At least in theory, this will lead to early treatment for some and peace of mind for others.
“This transforms the patient experience from prolonged anxiety to proactive management, which is critical in an era of evolving Alzheimer’s treatment where accurate prognosis can help identify appropriate treatment candidates,” Darmian said in a press release announcing the FDA approval. “BrainSee’s economic impact will be significant for all healthcare stakeholders and promises to reduce the billions of dollars spent annually on Alzheimer’s care through more effective management and treatment.”
of the FDA “De Novo” designation means that the product has no open market predecessors, but has proven its effectiveness and safety in clinical trials. BrainSee first received the FDA’s “breakthrough” designation in 2021, an earlier stage in the approval pathway for a first-of-its-kind treatment.
Darmiyan says BrainSee is fully automated and provides results the same day scans and cognitive tests are entered. The company sees the technology as changing the treatment of mild/early cognitive decline from biomarker-based methods to “non-invasive and actionable predictions of future improvement or progression.”